
(Note: you can also do this test on a solid sample). Using a pipette, collect the gas given off and bubble it through the limewater.Put your sample in a separate test tube and add a few drops of hydrochloric acid. 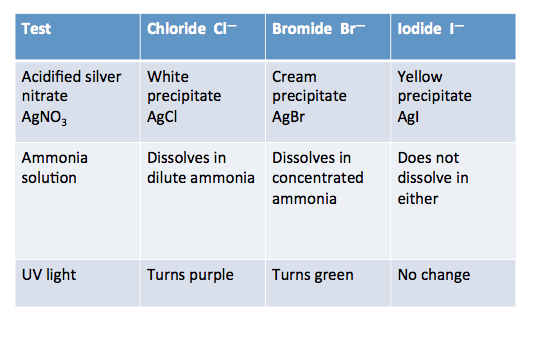
Put a small amount of limewater into a test (no more than 1 cm 3).Silver nitrate solution can stain skin and clothes. (See CLEAPSS Hazcard HC087).Nitric acid is an irritant. (See CLEAPSS Hazcard HC067).Keep the lid on the bottle when not in use. (See CLEAPSS Hazcard HC006) However, it can give off ammonia vapour, which can irritate the eyes and lungs. Ammonia solution is an irritant when concentrated, but not at the concentrations used by students in this activity.Wash your hands after use and warn students to do the same. Barium chloride solid is toxic the 0.1 mol dm −3 solution is harmful.Read our standard health and safety guidance.Carbonate solution, eg potassium carbonate.Sodium hydroxide solution less than 0.5 mol dm −3.To make the second part of the exercise more challenging, tests for positive ions could be introduced and students could be asked to identify both the positive and negative ions present in a solution. In the second, they use their observations to help them identify the negative ions present in a number of unknown solutions. This activity is in two parts – in the first, students make observations while carrying out the tests for various negative ions. Mix well, watch closely, and learn! Introduction Observation is key in this practical, as all the information needed will be seen in the reactions present. RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.Using a spatula, place a small amount of the solid containing halide ions into a test tube and then add a few drops of concentrated sulfuric acid.Halide Ions (Solid): Halide ions in solids can be identified through a reaction with concentrated sulfuric acid. For different halides, different coloured precipitates are formed. Halide Ions (Aqueous): Halide ions in an aqueous solution can be identified through a reaction with silver nitrate. If a white precipitate is formed, sulfate ions are present.In a test tube, mix an equal amount of the substance containing sulfate ions, dilute hydrochloric acidand barium chloride.Sulfate ions: Sulfate ions can be identified through a reaction with barium chloride. If the lime water goes cloudy, carbonate ions are present. Bubble the gasproduced through limewater. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
